




The man from Down Under : Archie Blue Archie Blue, from Christchurch, New Zealand, was both a professional athlete in his younger years and an avid inventor. In the 1970s he patented and demonstrated publicly and before experts a device that fit in the motor compartment of a car which converted water into combustible hydrogen and oxygen. He proved his invention on many occasions, and is featured in the book "Suppressed Inventions & Other Discoveries". He was offered big money for his invention, which he refused, and when he died of old age, his family discarded a "pile of junk" that had belonged to him at the local dump. As of the time of this writing, his invention has never been marketed Peter Lowrie, another New Zealander who actually met Mr. Blue in the 70's, moderates a Yahoo chat group (egaspower forum has mysteriously been taken down?, but we got a new one : hydroxy), developing water fuel devices inspired by Mr. Blue's design. The group is sharing information as they perfect their devices. Mr. Lowrie feels that burning fossil fuel has adversely affected our planet, and hopes his effort will help. info. from James Allen |
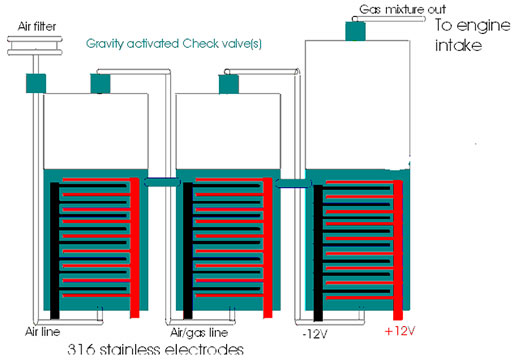
Here is a simple diagram of a 3 stage Archie Blue egas system by Peter Lowrie. The right voltage, amps, the right circuit, the right frequency, an IMPCO propane carb.(You can find them on ebay.) Air in coming from from your exhaust, going into a water tank instead of a gas tank. It will produce enough combustional egas to run a 4 cylinder ICE motor. (read below) Chemical engineers & physicist will tell you it will not work. It breaks the laws of physics and it simply will not work. Have they tired this themselves? They measure by BTU's (heat) while 78% of a gasoline ICE goes right out the exhaust pipe, in heat energy wasted. Hint: You don't need the heat, you just need combustion and egas produces enough in this system. Go make one it is not that hard! Open your mind and learn a new thing. |







